About our project

The problem we solve: The practice of suturing is centuries old & incredibly important for correct healing to occur. It seems like such a simple, common procedure that its proper practice & execution is often overlooked and taken for granted. Every year we fall short of the 300 million surgeries to meet the global disease burden. Shortage of trained surgeons, as well as accessibility & affordability are at the core of the problem. Suturing is an integral part of surgery that requires proficiency & is paramount to the
success of the surgical procedure. With the shortage of trained surgeons, the surgeons’ time & cost is at a premium. Devices that empower healthcare practitioners (surgeons’ assistants, nurses, or EMTs) to readily suture patients on the go, reliably and consistently, would have a positive impact on patient outcomes & thus their quality of life. Suturing remains a challenge to both experts and non-experts, and can lead to severe consequences if not done properly.

About our solution:
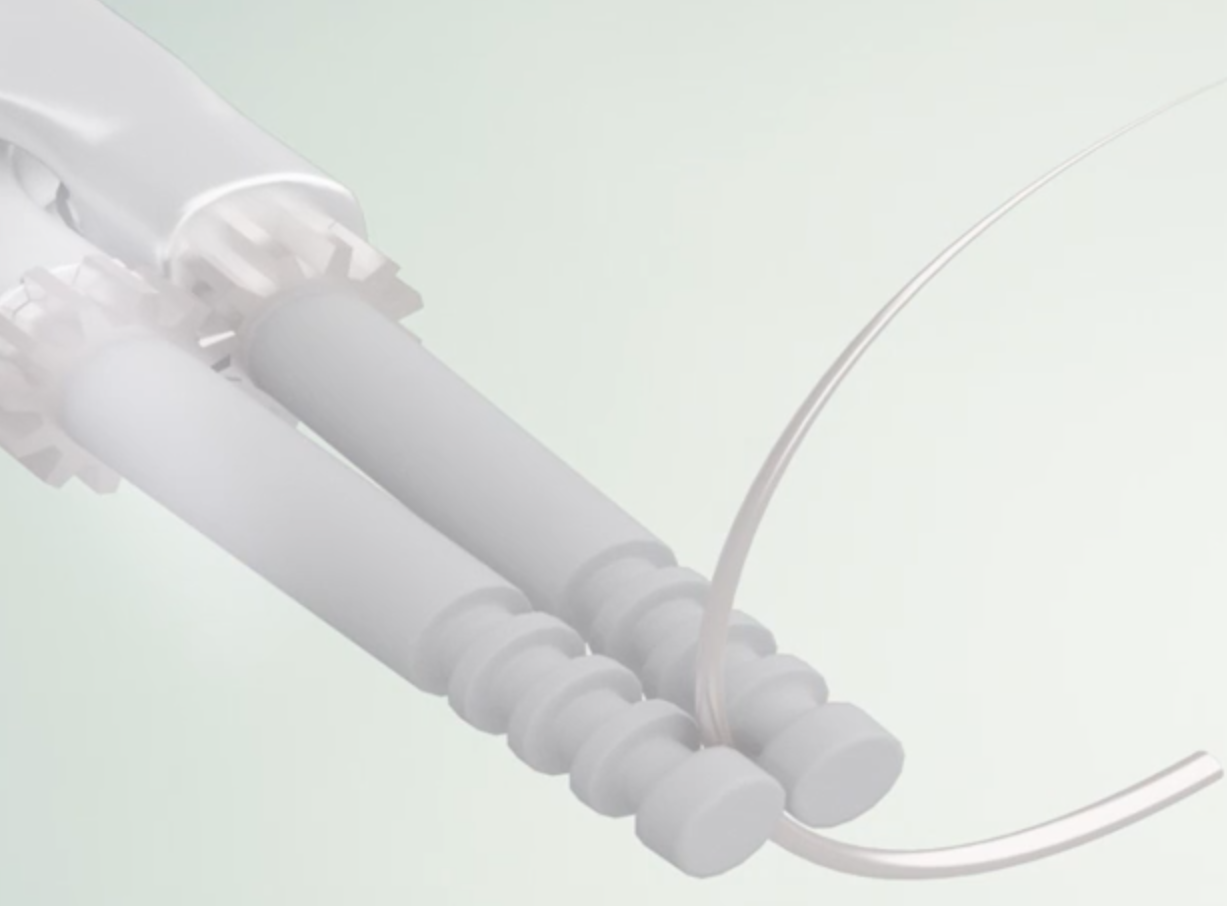
We design solutions to streamline the suturing process, limiting physical stress & hand-fatigue, preventing potential tearing of tissues & improving the end-user expertise, regardless of their suturing proficiency level, hence increasing patient safety. Ergosuture's "Drive'N Roll" is an enhanced suturing device allowing operator to effortlessly drive & roll a needle through tissues.
 Progress to
date:
Progress to
date:The final prototype design is currently in development for our reusable hand-held suturing device - The Drive'N Roll. Development of our disposable hand-held device prototype will commence in the next 2-3 months. We already have a nice pool of surgeons lined up to field-test and try our devices and the overall initial reaction from the Surgeon Community has been positive to say the least. 93% of Surgeons (n > 100) exposed to the Ergosuture Suturing Concept so far, believe we are bringing innnovation to the suturing space! We have also initiated our outreach (and gotten traction from large companies) for partnering in the development of the Laparoscopic & Robotic applications of the Ergosuture Platform.
About Our Team

Creator: Claude Nogard
Location: Massachusetts
Bio: I am a Global Marketing Strategy Expert with 20 years experience in US & international markets. I honed my skills & expertise at Merck & Novartis, working in various functions; brand management, market development & new product launches. I recently co-founded Ergosuture, with the goal to reinvent traditional suturing to meet today’s global surgical needs. Ergosuture is committed to developing suturing devices that simplify & expedite the suturing process for both expert & non-expert users.
Title: CEO
Advanced Degree(s): Masters Degree
About Team Members
Luis Almodovar
Neurosurgeon, MD
Biography: Specialties: Brain tumor resection, awake surgery for brain tumor resection, frameless stereotactic brain biopsy procedures, Gamma knife radiosurgery, Cyberknife radiosurgery, invention and development of medical devices
Title: Neurosurgeon
Advanced Degree(s): MD
Twitter:
@ERGOSUTURE
LinkedIn:
https://www.linkedin.com/in/luis-j-almodóvar-74a17342
About Our Company

How We Help Patients
Suturing with Ergosuture devices is less physically demanding on the Surgeon, reducing stress & hand-fatigue, thus limiting a main contributor to surgical incidents and patient post-surgical complications (infections & bleeding).
How We Help Physicians
We design solutions that streamline the suturing process, aimed at limiting physical stress & hand-fatigue of the surgeon, preventing potential tearing of tissues & thus improving the end-user experience, while increasing patient safety. Ergosuture's "Drive'N Roll" Technology is an enhanced suturing concept that allows the operator to effortlessly drive & roll a needle through tissues. The Ergosuture Technology Platform aims to close the global surgical needs gap, currently estimated at 130 million procedures per year.
How We Help Hospitals
The Ergosuture technology platform, with its enhanced suturing devices, will allow hospitals, clinics and medical institutions to make better use of its resources. By streamlining the suturing process and making wound closure easier, better and faster, valuable time is saved, allowing medical professionals (surgeons, nurses, etc) to have a higher # of patient-per rate on a daily basis. The improved wound closure productivity could also result in Surgeons being able to schedule additional surgeries on any particular day, thus eventually improving the bottom line of the medical facility.
How We Help Partners
The Ergosuture Suturing Technology can help current & future robotic, robot-assisted and/or laparoscopic surgery manufacturers differentiate their offering as well as enhancing their value proposition - enabling faster suturing in tighter spaces during minimally invasive procedures.
Innovation Details
Intellectual Property Summary
To date, Ergosuture has
–Three major patents obtained in the US, including for Laparoscopic & Robotic Suturing applications,
–One pending patent covering new trigger mechanism for re-usable Drive’N Roll,
–One provisional patent which covers multiple improvements of Drive’N Roll handheld, including needles & tip designs, & Drive’N Rock
Obtained Patent #'s: US 7,331,970 B2 / US 8,696,690 B2 / US 9,192,376 B2
Clinical Information
The early stage Ergosuture devices (hand-held devices) will most likely be designated by the FDA as Class-I (exempt) and does not require clinical trials.
Larger scale formal field-tests will be conducted in the future, utilizing partnering NGO's & ERGOSUTURE Surgeon's Club Members.
The clinical efficacy and required FDA filings/approvals for the Laparoscopic and Robotic Suturing applications will be pursued with our future licensing partner(s).
Regulatory Status
Ergosuture Handheld devices: Class-I (exempt) FDA-classification
878.4800 (Product Code HCF – Instrument, Ligature Passing and Knot tying)
Ergosuture Laparocscopic/Robotic Applications: Most probably Class-I 510(k) Non-Exempt FDA-classifications
How we will use the funds raised
The funds generated through this MedStartr campaign will primarily go towards product development, field-testing and emerging markets development activities. In addition, we would initiate feasibility studies of the 2nd generation Ergosuture device, the Drive'N Rock, which is particularly well-suited for high volume suturing scenarios; ER, Emergency Management, Field-Hospitals, Disaster Relief, Cosmetic & Reconstructive Surgeries. The Drive'N Rock will compete primarily against surgical staplers (and traditional suturing with needle drivers), but with the added benefit of being able to suture multiple layers - skin & facia (deeper tissues) with one device.
Thank You
We are surgeons. We are the ones that sacrificed long nights, plowed through endless work and faced quite difficult problems that only we had the skill set and experience to solve. We care for our patients in a very involved, direct and definitive way. All that effort has not only prepared us to better serve the ones who need us the most but has also resulted in an accumulation of vast amount practical surgical knowledge that nobody else can claim. That knowledge and continuous exposure to challenges and situations within and outside the OR has made us keenly aware of what we need to solve problems. I bet that most, if not all, of you have found yourself at one time or another saying "if I could have something that could do this or that to help me perform this task". That is exactly what happened to me. I realized that this technology could help us suture in situations in which suturing is difficult, time-consuming or tedious. Our suturing technology has been designed by a surgeon to serve the needs of surgeons and patients alike. By supporting our project you are not only supporting us, but also supporting all the surgeons that would benefit from using our devices. Faster and easier suturing also means less fatigue for you and less time spent in the OR. That is good for the patient on the OR table and for the patients waiting for you outside the OR as well, not to mention the institutional benefits too.
Please help us redefine suturing!
Dr. Luis J. Almodovar - Neurosurgeon
Inventor of the Ergosuture Technology Platform



 Progress to
date:
Progress to
date:














