GlucoSight: Blood Sugar at the Speed of Sight
At GlucoSight we are on a mission! Our goal is to reduce the daily burden of diabetes by developing a pain-free, low-cost means of monitoring blood sugar. No Lance! No Blood! No Pain! Just a glance!
Nashua, NH United States Diabetes BigData Medical Device MedStartr Ventures challengeAbout our project
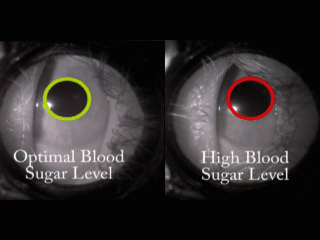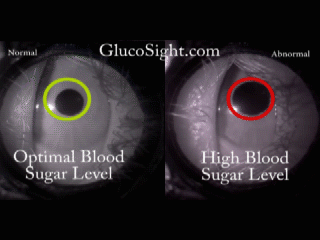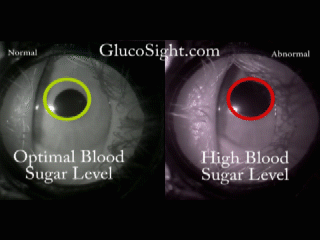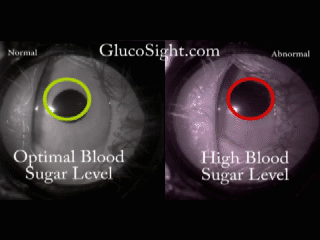
The problem we solve: Over 400 Million people in the world, those with Diabetes, have to inflict pain on themselves multiple times every day in order to live a healthy life. In addition to the pain, they are required to endure addition expense by having to continuously purchase strips and lances, adding insult to injury. Shouldn't there be a better way?

About our solution: Theory: The reaction of the pupil is a leading indicator of diabetic glucose levels Our solution employes two techniques: First: Placing the pupil in a light isolation environment, bathing the pupil with infrared light, and recording the pupillary reaction when we apply a short burst of stimulus (a white light). Second: Analyzing the video of the pupil reaction and compare the reaction of the pupil to previously derived, individualized baselines. The patient simply holds the GlucoSight to their eye for 8 seconds, we record the video, analyze the results and give them an indication as to whether their glucose level is healthy or not. There is a lot of lighting magic and proprietary software that we had to develop, but the pupillary reaction curve allows us to derive the patients glucose range.
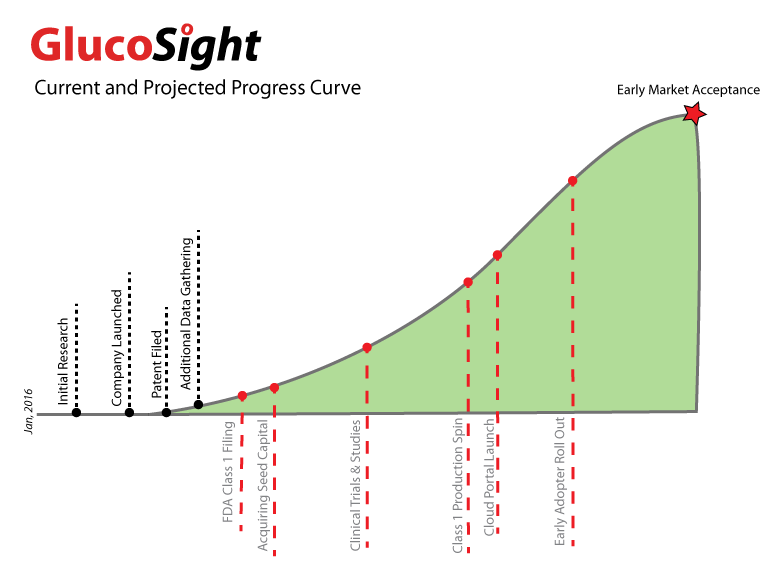
Progress to date:
At this point we are pre-market. The IP has been filed, data collection is ongoing, hardware and software are on continuous innovation/quality cycles, and paths to market both domestically and internationally, direct and partnerships are being actively pursued.
We are filing the initial paperwork for the FDA and will be launching a study with the Department of Endocrinology at a reputable Boston-based hospital and medical school in August and should have results very quickly as the GlucoSight feeds the resulting data into our cloud servers, allowing us immediate access to the data.
About Our Team

Creator: Ken Steinberg
Location: New Hampshire
Bio: A 30-year industry veteran with a reputation as a prolific problem solver with a continuously growing portfolio of solutions (products, services, patents) in numerous fields including computer science, healthcare and consumer safety.
Title: CEO/Founder
Advanced Degree(s): BSCS
About Team Members
Jason Clark
Founder/ VP HW Engineering, BSEE
Biography: 20 Year industry veteran with an extensive background in mechanican, electrical, and software engineering. Jason has deveoped numerous products ranging from medical devices to lighting to robotics.
Title: Founder/ VP HW Engineering
Advanced Degree(s): BSEE
LinkedIn:
https://www.linkedin.com/in/jasonclarknh
James Wilkie
Head of Regulatory Affairs, BS Plastics
Biography: A medical industry executive with extensive experience in M&A, R&D, regulatory affairs and operations in biotech, catheter and surgical product companies.
Title: Head of Regulatory Affairs
Advanced Degree(s): BS Plastics
LinkedIn:
https://www.linkedin.com/in/jim-wilkie-b974861
Dave Hall
Dir, Business Development, BSME
Biography: Dave Hall has over 30 years experience in Sales, Marketing and Business Development, working as a Sales and Business Consultant for Electronics, Medical Device, Bio-Medical, IT and Industrial accounts. A Mechanical Engineering graduate from the University of New Hampshire
Title: Dir, Business Development
Advanced Degree(s): BSME
LinkedIn:
https://www.linkedin.com/in/hallzy
John Brooks III
Advisor, MSBA
Biography: An industry stalwart with over two decades of work in diabetes including executive leadership at Joslin Diabetes Center, Pfizer, and Insulet.
Title: Advisor
Advanced Degree(s): MSBA
LinkedIn:
https://www.linkedin.com/in/john-brooks-ab04432
Dan Grunberg
Advisor, PhD EECS - MIT
Biography: Founder of and Chief Science Officer for Immedia Semiconductor and President of Chainwave Systems, Dan is well-know for his expertise in imaging.
Currently a principal at Apollo Partners specializing in health insurance services
Title: Advisor
Advanced Degree(s): PhD EECS - MIT
Dr Demetrios Vavvas
Advisor, M.D. Ph.D, Mass Eye and Ear
Biography: Demetrios Vavvas, M.D., Ph.D., is a full-time clinician scientist in the Retina Service of Mass. Eye and Ear, an Associate Professor of Ophthalmology with Harvard Medical School, a Principal Investigator in the Angiogenesis Laboratory, and the incumbent of the Monte J. Wallace Ophthalmology Chair in Retina.
Title: Advisor
Advanced Degree(s): M.D. Ph.D, Mass Eye and Ear
LinkedIn:
http://www.masseyeandear.org/find-a-physician/v/vavvas-demetrios
About Our Company

GlucoSight
Location: 356 Main Dunstable Rd
Nashua, NH 03062
US
Founded: 2016
Website: http://www.glucosight.com
Twitter: GlucoSight
Facebook: https://www.facebook.com/GlucoSight
Product Stage: Prototype/MVP
YTD Sales: Working on it
Employees: 5-10
How We Help Patients

Early detection and ongoing maintenance are crucial to the longevity and health of someone with diabetes. Many of the complications arise when the patient does not properly monitor their blood sugar as prescribed. Unfortunately the cost and pain associated with traditional lance-based methods act as a detractor, a deterrent to the patients daily routine monitoring requirements.
With a GlucoSight, we remove the negative (pain and cost) stimuli associated with monitoring one's blood sugar. We have had countless individuals ask us how to get a GlucoSight. They want relief, we all want to better outcomes, and a pain-free lifestyle.
How We Help Physicians
Do you wish your patients managed their diabetes more consistently and reliably? Do you wish you could remotely monitor their efforts AND experience the effects on the physiology? Are you concerned for your patient’s overall health due to their poor monitoring habits?
At GlucoSight, our intent is to drive better outcomes by making the process of monitoring blood glucose levels pain-free. Imagine the ability of a patient to simply look into a small, mobile-phone attached device and in matter of seconds know if they are hyper- or hypoglycemic. At the same time, these results, and physiological information, including actual video of their eyes, is readily available to you via your laptop or phone?
All of this is potentially possible with your donation. Help us make better diabetic outcomes a reality!
" placeholder="" maxlength="2000">
Do you wish your patients managed their diabetes more consistently and reliably? Do you wish you could remotely monitor their efforts AND experience the effects on the physiology? Are you concerned for your patient’s overall health due to their poor monitoring habits?
At GlucoSight, our intent is to drive better outcomes by making the process of monitoring blood glucose levels pain-free. Imagine the ability of a patient to simply look into a small, mobile-phone attached device and in matter of seconds know if they are hyper- or hypoglycemic. At the same time, these results, and physiological information, including actual video of their eyes, is readily available to you via your laptop or phone?
All of this is potentially possible with your donation. Help us make better diabetic outcomes a reality!
" placeholder="" maxlength="2000">

Do you wish your patients managed their diabetes more consistently and reliably? Do you wish you could remotely monitor their efforts AND experience the effects on the physiology? Are you concerned for your patient’s overall health due to their poor monitoring habits?
At GlucoSight, our intent is to drive better outcomes by making the process of monitoring blood glucose levels pain-free. Imagine the ability of a patient to simply look into a small, mobile-phone attached device and in matter of seconds know if they are hyper- or hypoglycemic. At the same time, these results, and physiological information, including actual video of their eyes, is readily available to you via your laptop or phone?
All of this is potentially possible with your donation. Help us make better diabetic outcomes a reality!
How We Help Hospitals

We know every member of your team cares about the health of your patients, but we also know it is a daily challenge to keep the cost of healthcare in check. We are here to help.
With Medicare reimbursement cuts on the horizon, we can help reduce the cost of monitoring glucose levels and achieve better outcomes for your diabetic patients by eliminating the costs of lances, strips, excessive lab tests, and additional services due to poor glucose monitoring. With an 8 second video, we can help your staff work with your patients to easily and effectively monitor their diabetes. The time, cost, and long term complications that your hospital deals with every day might become a thing of the past.
Donate and help us, help you!
How We Help Partners
Imagine how GlucoSight effects all three of these areas; we can screen earlier and faster with less education; we can remove the costs of lances, strips, nursing time; we can increase outcomes due to easier monitoring. All with a simple mobile-device, a cellphone, and software, but with none of the headaches that go with producing, packing, and shipping sterile medical devices.
Add to this cloud-based access to patient data and data analytics. Every data element and image collected by GlucoSight securely resides in the cloud where healthcare professionals can provide ongoing treatment adjustments. Not only does the patient benefit but so does their physician and fellow researchers.
The market is changing. Companies, like GlucoSight, are racing for better, less costly, less painful, monitoring tools while others are striving for better treatment with innovations like the artificial pancreas. With tens of billions of dollars on the line, companies must either innovate or watch others cut into their bottom line. GlucoSight is poised to be a technology that will allow existing (or potential) market players to stay in the game by offering less expensive solutions to their customers, and better data to their researchers.
Imagine how GlucoSight effects all three of these areas; we can screen earlier and faster with less education; we can remove the costs of lances, strips, nursing time; we can increase outcomes due to easier monitoring. All with a simple mobile-device, a cellphone, and software, but with none of the headaches that go with producing, packing, and shipping sterile medical devices.
Add to this cloud-based access to patient data and data analytics. Every data element and image collected by GlucoSight securely resides in the cloud where healthcare professionals can provide ongoing treatment adjustments. Not only does the patient benefit but so does their physician and fellow researchers.
The market is changing. Companies, like GlucoSight, are racing for better, less costly, less painful, monitoring tools while others are striving for better treatment with innovations like the artificial pancreas. With tens of billions of dollars on the line, companies must either innovate or watch others cut into their bottom line. GlucoSight is poised to be a technology that will allow existing (or potential) market players to stay in the game by offering less expensive solutions to their customers, and better data to their researchers.
" rows="4" placeholder="" maxlength="2000">

Imagine how GlucoSight effects all three of these areas; we can screen earlier and faster with less education; we can remove the costs of lances, strips, nursing time; we can increase outcomes due to easier monitoring. All with a simple mobile-device, a cellphone, and software, but with none of the headaches that go with producing, packing, and shipping sterile medical devices.
Add to this cloud-based access to patient data and data analytics. Every data element and image collected by GlucoSight securely resides in the cloud where healthcare professionals can provide ongoing treatment adjustments. Not only does the patient benefit but so does their physician and fellow researchers.
The market is changing. Companies, like GlucoSight, are racing for better, less costly, less painful, monitoring tools while others are striving for better treatment with innovations like the artificial pancreas. With tens of billions of dollars on the line, companies must either innovate or watch others cut into their bottom line. GlucoSight is poised to be a technology that will allow existing (or potential) market players to stay in the game by offering less expensive solutions to their customers, and better data to their researchers.
Challenge Mission
Market Size
Focusing on the US market initially, we have had in-depth conversations with some of the distribution arms for the largest existing diabetes glucose meter "Big Box Retail/OEM" distributors in order to understand the scale and opportunity of the market.
Based on their numbers, the US market consists of 20M glucose meters. Every year 20% of that market refreshes, meaning diabetics get new meters due to meter failures, new technology etc. This is approximately 4M meters a year.
In order to determine a what percentage of these 4M units we could potentially capture annually, we ran a Facebook add for 45 days (see image) asking the Diabetic community if they would rather "Lance or Glance". In 45 days we got 84K impressions with a CTR (click thru rate) of 3.7%. Applying this rate to the 4M available units would equate to a reachable market of 150K units annually.
Projected 3 Year Growth
We have performed a 5 year detailed, P&L projection based on the Diabetes as a Service model which provides us with reocurring revenue of $60 per person per year. With a capture rate of 150K unit per year ($9M in annual revenue per 150K) we conservatively project US only revenue to be $27M by 2020 with an EBITDA of $12M.
How We Will Make Money
Given that our solution does not require the use of strips or lances this allows us the flexibilty to provide our solution at market disrupting prices. We are going to offer our solution as a service, Diabetes As A Service (DaaS). The consumer will sign up for the service for an initial fee of $9.99 and then pay $4.99 per month ($60 per year), unlimited usage.
In additional we will charge care givers and physicians access to our cloud ports at a discounted monthly rate and will generate additional revenue via advertizements on our mobile application.
We have also engaged one of the leading OEM/Big Box Retail distribution firms which will be handling the OEM and Retail strategy. This firm has over 10 years of experience selling into the existing retailers for firms such as DexCom, J&J, GE and others. They have all of the relationships necessary to bring an OEM product to market in under a year.
About our Competition
There are no companies that currently offer our technology or solution. There are a number of academic institutions which are working on products that analyze the chemical properties of tears and exhaled gases. These solutions require either implated devices or chemical analysis strips. We are the only company which is performing image analysis to determine Glucose range.
Innovation Details
Intellectual Property Summary
A 24-page provisional patent has been filed covering the method for pupillary process analysis. Further Design patents and trademarks are in process. Additional prior art searches are being conducted for image analysis methods.
Clinical Information
Currently we are collecting data on a daily basis from new participants. We send GlucoSight units out to volunteers who use the GlucoSight in concert with their blood or continuous glucose monitor. These volunteers record their blood glucose level and video via our web application which submits this non-attributed, secure data to our data server. This process of data collection is ongoing.
In addition, we are engaging with clinical researchers at various medical and university institutions to engage in more detailed analysis and study. We expect to have additional studies in place in the Fall of 2016.
Regulatory Status
We have recently filed our 510K pre-Submission for a combination Class 1/Class 2 approval. Our solution consists of a Class 1 exempt hand-held pupilometer and a Class 2 mobile software application that performs the analysis.
We expect to meet with the FDA in January 2017 with approval by the 3rd quarter of 2017.
How we will use the funds raised
The funds will be used to conduct a 30 person trial at one of the leading diabetes research centers in Boston, MA, USA. We have spoken with a number of facilities which are prepared to conduct this study at a cost equal to or less than the capital we are seeking.
Thank You
We want people with diabetes to live longer and have a healthy lifestyle without complications. We feel the best way to support this goal is to make the process of monitoring one's glucose level as painless and easy as possible. What could be easier that looking into a small camera for 8 seconds and having a result pop up on your phone?
This is our goal. We all have loved ones who deal with diabetes and we know there is a better way. We decided not to let the market come to us but to introduce our own solution with the intent of introducing market change and ultimately better outcomes.

Updates
No updates found .
Supporters
-

11/25/2016 - Liked the project.
11/23/2016 - Liked the project.
11/23/2016 - Liked the project.
11/22/2016 - Liked the project.
11/22/2016 - Liked the project.
11/21/2016 - Liked the project.
11/21/2016 - Liked the project.
11/21/2016 - Liked the project.
11/21/2016 - Liked the project.
11/20/2016 - Liked the project.
11/20/2016 - Liked the project.
11/20/2016 - Liked the project.
11/20/2016 - Liked the project.
11/20/2016 - Liked the project.
11/20/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/19/2016 - Liked the project.
11/18/2016 - Liked the project.
11/18/2016 - Liked the project.Instant Feedback
Help us find best new ideas to fund by telling us what you think. Your feedback goes straight to the team behind this project in private, so tell them what you really think.
90Medstartr
Index Score90
Interest
Score0
Adoption
Score30
Likes0
Partners0
Pilots0
Follows-
This campaign has ended but you can still get involved.See options below.
$ 85,000 goal
Instant Feedback
Help us find best new ideas to fund by telling us what you think. Your feedback goes straight to the team behind this project in private, so tell them what you really think.




